 The Van Der Waals equation is a state that shows two properties of gases, such as the excluded volume of real gases and its attractive forces. Some Van Der Waals forces examples are hydrogen bonding, dipole-dipole interactions, and dispersion forces. Further, they are known as a universal interaction between various particles, divided by mediums of air or vacuum. However, they also involve a repulsive interaction within molecules, arising from the overlapping of more than two atomic electronic clouds situated closer to each other. The Van Der Waal forces include attractions within various atoms, resulting from influenced dipoles. In comparison to most of the ionic and covalent bonds, these interactions do not arise from an electronic bond. In solids, these forces hold each other having lower melting points, and are relatively softer than the ones held by ionic or metallic bonds. In 1873, he first discovered the Van Der Waals bond while working on a theory on real gasses.Īccording to the Van Der Waals forces definition, they are comparatively weaker electrostatic forces that attract uncharged or neutral molecules towards each other in almost all organic liquids, gasses, and solids. 
Moreover, they are the weakest intermolecular forces, consisting of dipole-dipole and dispersion forces.Īs a segment of molecular physics, these forces came into existence from the name of a Dutch Scientist, Johannes Diderik Van Der Waals. These interactions include weak electrostatic forces lying in a close range within molecules lacking charges. StudySmarter Original.In simple words, Van Der Waals Forces are those bonds that play the role of attracting both molecules and atoms. 
The movement of electrons leads to a temporary dipole. Let's start by looking at what happens when a temporary dipole is formed. 
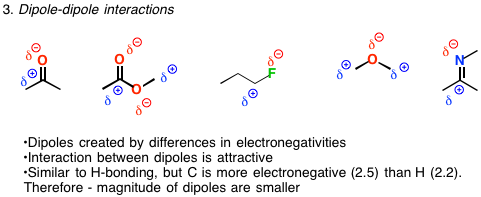
London dispersion forces are due to two types of dipoles: temporary and induced. To better understand these definitions, let's look at some diagrams. Where, the solid dashes, -, represent covalent bonds, and the dots, …, represent a hydrogen bond.ĭipole-dipole Force - an attractive intermolecular force that causes molecules that contain permanent dipoles to align end-to-end, so that the positive end of a given dipole on one molecule interacts with the negative end of a dipole on an adjacent molecule.Ĭovalent Bond - a chemical bond in which electrons are shared between atoms.Įlectronegativity - a measure of the ability of a given atom to attract electrons to itself. Hydrogen bonds are weaker (range: 10 kJ/mol - 40 kJ/mol) than covalent bonds (range: 209 kJ/mol - 1080 kJ/mol) and ionic bonds (range: lattice energy - 600 kJ/mol to 10,000 kJ/mol) but stronger than intermolecular interactions. Hydrogen Bond - attractive force between a strongly electronegative atom, X, bonded to a hydrogen atom, H, and a lone pair of electrons on another small, electronegative atom, Y.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
